JESEI
teacher’s notes
student’s notes




Carbon cycle: exchanging carbon dioxide between the atmosphere and
ocean (teacher’s notes)
This material is designed for students aged 12 to 14 (years 8 and 9).
Topic
This activity is one of five aimed to teach students about the nature of carbon, the different types of
compounds it exists in (eg charcoal, glucose, carbon dioxide), the biochemical reactions it takes part in
(photosynthesis and respiration), the range of processes that carbon and carbon compounds are involved in
on Earth, and how these link together form the carbon cycle.
The other activities are
Carbon cycle: where is this crucial carbon?; a teacher-led discussion interspersed with demonstrations in
which the teacher burns a range of materials over a Bunsen flame, forming charcoal, to illustrate that they
contain carbon.
Carbon cycle in the lab: carbon products and the processes that link them; which involves working in small
groups to place examples of the carbon cycle products in the correct places on a diagram of the cycle.
Cycling carbon: seeing how plants use carbon dioxide in the lab; a short pupil practical exploring the uptake
of carbon dioxide from the atmosphere by plants for photosynthesis.
Carbon cycle: releasing dinosaur breath in the lab; a short pupil practical exploring the storage of carbon in
the fossils that make up limestone and chalk.
Context
An understanding of the carbon cycle is essential to the debate about global warming, an environmental
issue that most students will have heard about. Since the Earth’s atmosphere formed, it seems to have
always contained carbon dioxide in varying amounts Carbon dioxide is a ‘greenhouse gas’ through which
light radiation can pass but which absorbs some of the heat radiation produced by light irradiating the
Earth’s surface. This causes the Earth’s surface and atmosphere to be warmer than it would otherwise be
and without the ‘greenhouse effect’ the Earth would probably be completely frozen. Humans, as all life on
Earth, have always been part of the carbon cycle, but now (since the industrial revolution) the large scale
burning of oil, coal and natural gas, along with deforestation, is leading to increasing atmospheric carbon
dioxide levels. This in turn is related to an enhanced greenhouse effect and consequent climatic change. An
understanding of the factors affecting global warming leads to an understanding of the measures required
to reduce their impact. This can link into economic and political debates on the subject. Students should
have already covered photosynthesis and respiration in order to do this activity effectively.
Teaching points
There is an exchange of carbon dioxide between the atmosphere and the ocean’s surface. Carbon dioxide
dissolves in the ocean and provides the source of carbon dioxide that plants and plankton living in the ocean
rely on for photosynthesis. The amount of carbon dioxide the ocean can contain depends on the
temperature of the water and on its saltiness (whether it is sea water or fresh water). Warm water can hold
more carbon dioxide in solution than cold water. When carbon dioxide dissolves in water, it forms carbonic
acid which makes the water acidic. In the lab we can test for the acidity caused by the presence of dissolved
carbon dioxide using Universal Indicator, which turns yellow when the solution is acidic. This activity tests
whether sea water or fresh water absorbs more carbon dioxide. Sea water can absorb more carbon dioxide
than fresh water without having major environmental effects.
Apparatus and materials
Each group of pupils will need:
Eye protection
2 beakers
Universal Indicator solution
Sea water (If real sea water is unavailable, a substitute can be made by dissolving approximately 30 g of
sodium chloride in 1 dm3 of water.)
Tap water (fresh water)
Drinking straw
Stopwatch
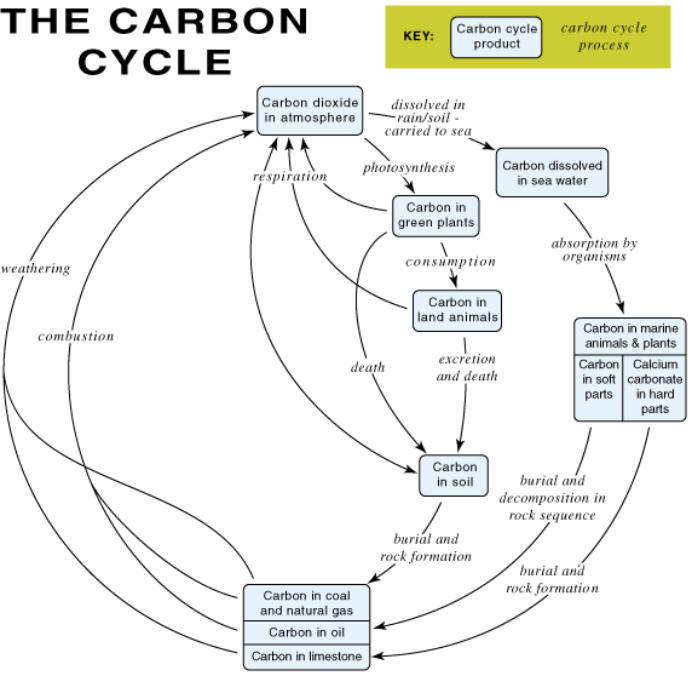
A copy of the carbon cycle diagram (Figure 1)
Safety
Pupils should wear eye protection, blow gently and not suck up the water. Dispose of straws at the end of
the activity.
Activity
The students should work in pairs or small groups. They pour 100 cm3 of sea water into one beaker and 100
cm3 of fresh water into the other. Then they add several drops of Universal Indicator to each so that the
colour is clearly visible. Next, using the straw, they blow gently and consistently into the water samples - first
the sea water, then the fresh water. For each they time how long it takes the indicator to become yellow and
record the results.
When this is done, the students should answer the following questions:
Q 1. What did it mean when the indicator was yellow? = That carbon dioxide had dissolved in the water to
produce an acid.
Q 2. Which beaker of water turned yellow the quickest? = The beaker of fresh water.
Q 3. Which absorbs more carbon dioxide without its acidity changing? = Sea water.
Q 4. Highlight this part of the carbon cycle on your diagram. = The arrows indicating exchange of gases at
the surface of the ocean should be highlighted.
The following questions concern the whole of the carbon cycle topic.
Overall questions
Q 1. Carbon is in the cycle in solid, liquid and gas forms. Which products show each of these forms (give
one example of each)? = For example; atmosphere – CO2, sea water, dissolved hydrogencarbonate ions
(HCO3-), coal – almost solid carbon, or limestone - CaCO3.
Q 2. Which processes happen quickly (give examples)? Which ones happen very slowly (give examples)? =
For example; quick processes – respiration, combustion, slow processes - limestone and coal formation.
Q 3. Which processes are going on outside the window today? = For example; photosynthesis, respiration,
CO2 dissolving in rain (if it is raining), weathering, consumption, excretion and death.
Q 4. Which processes do you take part in? = Respiration, consumption, excretion and death.
Q 5. Which processes did dinosaurs take part in? = Respiration, consumption, excretion and death plus
sometimes burial and rock-formation.
Q 6. Coal and natural gas formed from ancient plants. What processes affected these plants that probably
won’t affect the plants you see outside the window? = The plants outside are unlikely to become buried and
formed into rock – most plant materials rot away in the soil.
Figure 1 The carbon cycle

teacher’s notes
student’s notes




Carbon cycle: exchanging
carbon dioxide between the
atmosphere and ocean
(teacher’s notes)
This material is designed for students aged 12 to 14 (years
8 and 9).
Topic
This activity is one of five aimed to teach students about
the nature of carbon, the different types of compounds it
exists in (eg charcoal, glucose, carbon dioxide), the
biochemical reactions it takes part in (photosynthesis and
respiration), the range of processes that carbon and
carbon compounds are involved in on Earth, and how
these link together form the carbon cycle.
The other activities are
Carbon cycle: where is this crucial carbon?; a teacher-led
discussion interspersed with demonstrations in which the
teacher burns a range of materials over a Bunsen flame,
forming charcoal, to illustrate that they contain carbon.
Carbon cycle in the lab: carbon products and the
processes that link them; which involves working in small
groups to place examples of the carbon cycle products in
the correct places on a diagram of the cycle.
Cycling carbon: seeing how plants use carbon dioxide in
the lab; a short pupil practical exploring the uptake of
carbon dioxide from the atmosphere by plants for
photosynthesis.
Carbon cycle: releasing dinosaur breath in the lab; a short
pupil practical exploring the storage of carbon in the
fossils that make up limestone and chalk.
Context
An understanding of the carbon cycle is essential to the
debate about global warming, an environmental issue
that most students will have heard about. Since the
Earth’s atmosphere formed, it seems to have always
contained carbon dioxide in varying amounts Carbon
dioxide is a ‘greenhouse gas’ through which light radiation
can pass but which absorbs some of the heat radiation
produced by light irradiating the Earth’s surface. This
causes the Earth’s surface and atmosphere to be warmer
than it would otherwise be and without the ‘greenhouse
effect’ the Earth would probably be completely frozen.
Humans, as all life on Earth, have always been part of the
carbon cycle, but now (since the industrial revolution) the
large scale burning of oil, coal and natural gas, along with
deforestation, is leading to increasing atmospheric carbon
dioxide levels. This in turn is related to an enhanced
greenhouse effect and consequent climatic change. An
understanding of the factors affecting global warming
leads to an understanding of the measures required to
reduce their impact. This can link into economic and
political debates on the subject. Students should have
already covered photosynthesis and respiration in order
to do this activity effectively.
Teaching points
There is an exchange of carbon dioxide between the
atmosphere and the ocean’s surface. Carbon dioxide
dissolves in the ocean and provides the source of carbon
dioxide that plants and plankton living in the ocean rely
on for photosynthesis. The amount of carbon dioxide the
ocean can contain depends on the temperature of the
water and on its saltiness (whether it is sea water or fresh
water). Warm water can hold more carbon dioxide in
solution than cold water. When carbon dioxide dissolves
in water, it forms carbonic acid which makes the water
acidic. In the lab we can test for the acidity caused by the
presence of dissolved carbon dioxide using Universal
Indicator, which turns yellow when the solution is acidic.
This activity tests whether sea water or fresh water
absorbs more carbon dioxide. Sea water can absorb more
carbon dioxide than fresh water without having major
environmental effects.
Apparatus and materials
Each group of pupils will need:
Eye protection
2 beakers
Universal Indicator solution
Sea water (If real sea water is unavailable, a substitute can
be made by dissolving approximately 30 g of sodium
chloride in 1 dm3 of water.)
Tap water (fresh water)
Drinking straw
Stopwatch
A copy of the carbon cycle diagram (Figure 1)
Safety
Pupils should wear eye protection, blow gently and not
suck up the water. Dispose of straws at the end of the
activity.
Activity
The students should work in pairs or small groups. They
pour 100 cm3 of sea water into one beaker and 100 cm3
of fresh water into the other. Then they add several drops
of Universal Indicator to each so that the colour is clearly
visible. Next, using the straw, they blow gently and
consistently into the water samples - first the sea water,
then the fresh water. For each they time how long it takes
the indicator to become yellow and record the results.
When this is done, the students should answer the
following questions:
Q 1. What did it mean when the indicator was yellow? =
That carbon dioxide had dissolved in the water to produce
an acid.
Q 2. Which beaker of water turned yellow the quickest?
= The beaker of fresh water.
Q 3. Which absorbs more carbon dioxide without its
acidity changing? = Sea water.
Q 4. Highlight this part of the carbon cycle on your
diagram. = The arrows indicating exchange of gases at the
surface of the ocean should be highlighted.
The following questions concern the whole of the carbon
cycle topic.
Overall questions
Q 1. Carbon is in the cycle in solid, liquid and gas forms.
Which products show each of these forms (give one
example of each)? = For example; atmosphere – CO2, sea
water, dissolved hydrogencarbonate ions (HCO3-), coal –
almost solid carbon, or limestone - CaCO3.
Q 2. Which processes happen quickly (give examples)?
Which ones happen very slowly (give examples)? = For
example; quick processes – respiration, combustion, slow
processes - limestone and coal formation.
Q 3. Which processes are going on outside the window
today? = For example; photosynthesis, respiration, CO2
dissolving in rain (if it is raining), weathering,
consumption, excretion and death.
Q 4. Which processes do you take part in? = Respiration,
consumption, excretion and death.
Q 5. Which processes did dinosaurs take part in? =
Respiration, consumption, excretion and death plus
sometimes burial and rock-formation.
Q 6. Coal and natural gas formed from ancient plants.
What processes affected these plants that probably won’t
affect the plants you see outside the window? = The
plants outside are unlikely to become buried and formed
into rock – most plant materials rot away in the soil.
Figure 1 The carbon cycle


- Home
- contents
- help
- glossary
- Magnetic patterns 1
- Magnetic patterns 2
- Mantle convection
- Metamorphics
- Minerals & elements
- Plate riding
- Plate tectonic story
- Protecting the earth
- Rock cycle in lab
- Sedimentary rocks
- Separating mixtures
- Sequencing of rocks
- Solid mantle
- Structure of earth 1
- Structure of earth 2
- Structure of earth 3
- Tree rings
- Weathering
- Gravestones
- Lab volcano
- Investigate earth